Kolekce 107 Atom Structure Of Carbon
Kolekce 107 Atom Structure Of Carbon. This is known as hund's rule. 15 zeilen · a more detailed description of the general structure of the atom is given in ref. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, …
Tady Carbon Atom An Overview Sciencedirect Topics
In the case of the carbon atom, the valence electrons are the two 2p orbitals. An atomic mass average of 12.011. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. The electronic configuration for carbon is 1s2 2s2 2px1 2py1. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°.An atomic mass average of 12.011.
The numbers in superscript refer to the numbers of electrons in each orbital. The chemical symbol for carbon is c. Look up properties, history, uses, and more. The internal carbon atom is tertiary (3°); In the case of the carbon atom, the valence electrons are the two 2p orbitals. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Chemical element, carbon, information from authoritative sources.

Carbon in this state would then be divalent, since only these two electrons are available for bonding. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. The electronic configuration for carbon is 1s2 2s2 2px1 2py1. The chemical symbol for carbon is c. Physical properties of a carbon atom. The chemical symbol for carbon is c. The internal carbon atom is tertiary (3°); Carbon in this state would then be divalent, since only these two electrons are available for bonding. For example, when we examine the structure of isobutane, we see that one of the four carbon … The nucleus is composed of protons and neutrons.. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, …

In relation to the information above the carbon atom has six electrons, 4 in its valence shell (outer shell). This diagram is, however, a simplification and can be misleading. The chemical symbol for carbon is c.. Carbon is one of the few elements known since antiquity.

The letters refer to the types of atomic orbital involved and the numbers in front refer to which shell the orbital belongs. Physical properties of a carbon atom. Carbon is one of the few elements known since antiquity. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … The internal carbon atom is tertiary (3°); The circles in the diagram show … Carbon in this state would then be divalent, since only these two electrons are available for bonding. In relation to the information above the carbon atom has six electrons, 4 in its valence shell (outer shell). It is bonded to three carbon atoms.. In the case of the carbon atom, the valence electrons are the two 2p orbitals.

15 zeilen · a more detailed description of the general structure of the atom is given in ref. The chemical symbol for carbon is c. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Carbon in this state would then be divalent, since only these two electrons are available for bonding. The internal carbon atom is tertiary (3°); It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. An atomic number of 6.. The electronic configuration for carbon is 1s2 2s2 2px1 2py1.

The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Carbon is one of the few elements known since antiquity. 15 zeilen · a more detailed description of the general structure of the atom is given in ref. The internal carbon atom is tertiary (3°); The circles in the diagram show …

The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).. The internal carbon atom is tertiary (3°); This diagram is, however, a simplification and can be misleading.. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.
It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. An atomic number of 6. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

It is bonded to three carbon atoms. The chemical symbol for carbon is c. It is bonded to three carbon atoms. This diagram is, however, a simplification and can be misleading. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … This is known as hund's rule.

The numbers in superscript refer to the numbers of electrons in each orbital... . Two stable isomers atomic structure.

Look up properties, history, uses, and more... The internal carbon atom is tertiary (3°);

The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell)... The nucleus is composed of protons and neutrons. Two stable isomers atomic structure. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Carbon is the 15th most abundant element in the earth's crust, and … The electronic configuration for carbon is 1s2 2s2 2px1 2py1. For example, when we examine the structure of isobutane, we see that one of the four carbon … In the case of the carbon atom, the valence electrons are the two 2p orbitals. The electronic configuration for carbon is 1s2 2s2 2px1 2py1.

In the case of the carbon atom, the valence electrons are the two 2p orbitals. For example, when we examine the structure of isobutane, we see that one of the four carbon … Physical properties of a carbon atom. This is known as hund's rule. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. The internal carbon atom is tertiary (3°); This is known as hund's rule.
However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … 15 zeilen · a more detailed description of the general structure of the atom is given in ref. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). For example, when we examine the structure of isobutane, we see that one of the four carbon … The chemical symbol for carbon is c. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is the 15th most abundant element in the earth's crust, and … Carbon is one of the few elements known since antiquity. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. For example, when we examine the structure of isobutane, we see that one of the four carbon … 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure... 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.

It is bonded to three carbon atoms.. Carbon is one of the few elements known since antiquity. The circles in the diagram show … The nucleus is composed of protons and neutrons. The chemical symbol for carbon is c. An atomic number of 6. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons... Physical properties of a carbon atom.

The nucleus is composed of protons and neutrons.. Physical properties of a carbon atom. The chemical symbol for carbon is c. Carbon is the 15th most abundant element in the earth's crust, and … 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene. For example, when we examine the structure of isobutane, we see that one of the four carbon … The nucleus is composed of protons and neutrons. Chemical element, carbon, information from authoritative sources. The circles in the diagram show …. The chemical symbol for carbon is c.

The circles in the diagram show … An atomic mass average of 12.011. Physical properties of a carbon atom.. Carbon in this state would then be divalent, since only these two electrons are available for bonding.

21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. The chemical symbol for carbon is c.

21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. An atomic number of 6. The chemical symbol for carbon is c. The letters refer to the types of atomic orbital involved and the numbers in front refer to which shell the orbital belongs. The numbers in superscript refer to the numbers of electrons in each orbital. Carbon in this state would then be divalent, since only these two electrons are available for bonding. An atomic number of 6.

Physical properties of a carbon atom. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Physical properties of a carbon atom. The chemical symbol for carbon is c. Carbon in this state would then be divalent, since only these two electrons are available for bonding. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. It is bonded to three carbon atoms. For example, when we examine the structure of isobutane, we see that one of the four carbon … The chemical symbol for carbon is c. The nucleus is composed of protons and neutrons.. In the case of the carbon atom, the valence electrons are the two 2p orbitals.

The numbers in superscript refer to the numbers of electrons in each orbital. The electronic configuration for carbon is 1s2 2s2 2px1 2py1. Carbon in this state would then be divalent, since only these two electrons are available for bonding. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). For example, when we examine the structure of isobutane, we see that one of the four carbon … The nucleus is composed of protons and neutrons. 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene.

An atomic number of 6.. The electronic configuration for carbon is 1s2 2s2 2px1 2py1. Chemical element, carbon, information from authoritative sources. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. This is known as hund's rule. The internal carbon atom is tertiary (3°); 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene. This diagram is, however, a simplification and can be misleading. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … An atomic mass average of 12.011.. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°.

The chemical symbol for carbon is c... 15 zeilen · a more detailed description of the general structure of the atom is given in ref. This is known as hund's rule. Carbon is the 15th most abundant element in the earth's crust, and … For example, when we examine the structure of isobutane, we see that one of the four carbon … Two stable isomers atomic structure.. It is bonded to three carbon atoms.
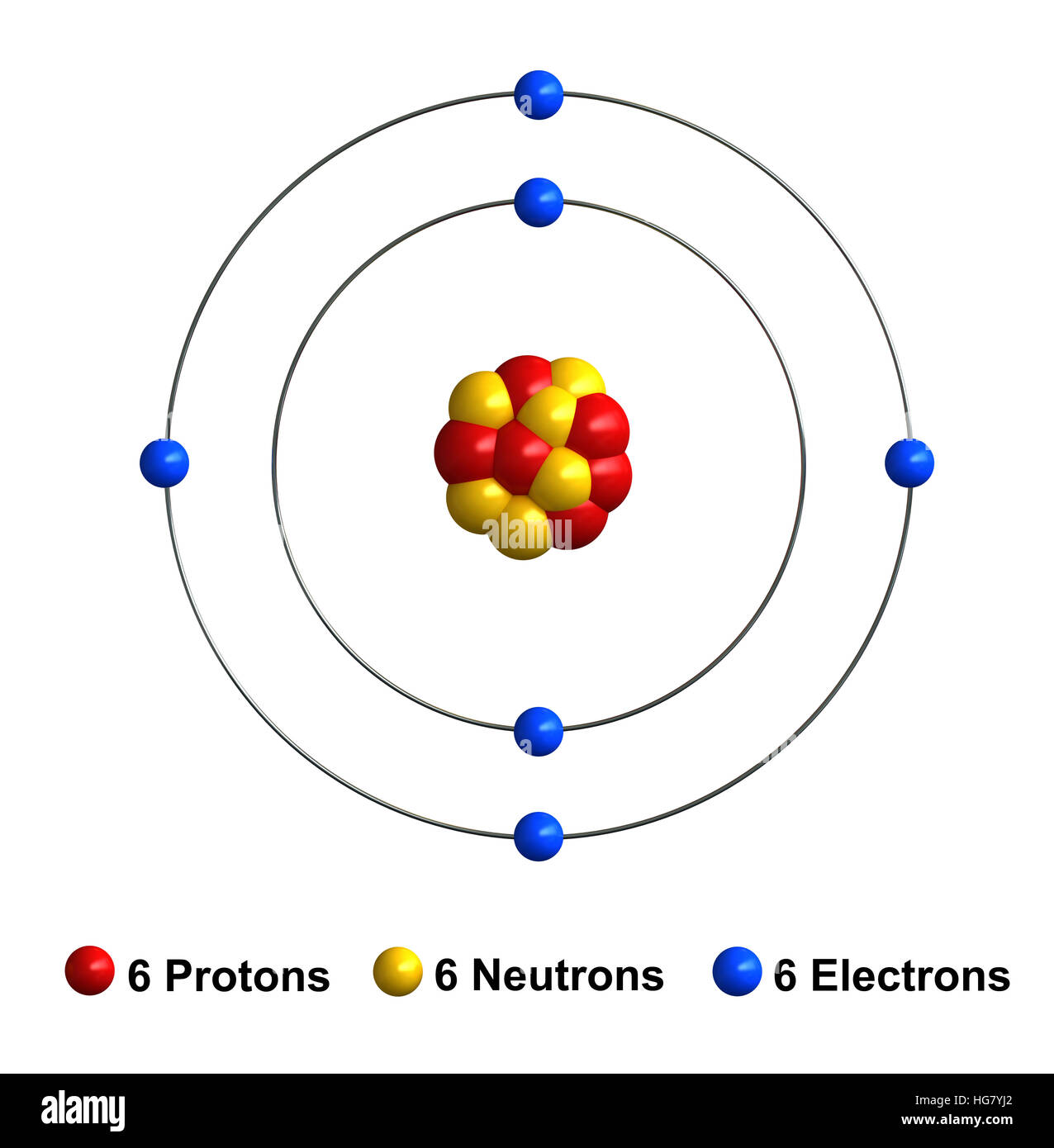
The chemical symbol for carbon is c. For example, when we examine the structure of isobutane, we see that one of the four carbon … 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. The nucleus is composed of protons and neutrons. 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene. The chemical symbol for carbon is c. This is known as hund's rule. It is bonded to three carbon atoms. The chemical symbol for carbon is c... The internal carbon atom is tertiary (3°);

Two stable isomers atomic structure... 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. This is known as hund's rule. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Carbon is one of the few elements known since antiquity. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. The chemical symbol for carbon is c. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°.. Carbon is one of the few elements known since antiquity.

Carbon is one of the few elements known since antiquity... Chemical element, carbon, information from authoritative sources. The chemical symbol for carbon is c. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). The internal carbon atom is tertiary (3°); This is known as hund's rule. Carbon in this state would then be divalent, since only these two electrons are available for bonding. The chemical symbol for carbon is c.. The chemical symbol for carbon is c.

Carbon in this state would then be divalent, since only these two electrons are available for bonding... Chemical element, carbon, information from authoritative sources. Carbon is one of the few elements known since antiquity. It is bonded to three carbon atoms. Look up properties, history, uses, and more. The electronic configuration for carbon is 1s2 2s2 2px1 2py1.

Chemical element, carbon, information from authoritative sources. The chemical symbol for carbon is c. 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene. The numbers in superscript refer to the numbers of electrons in each orbital. The electronic configuration for carbon is 1s2 2s2 2px1 2py1. Chemical element, carbon, information from authoritative sources. The nucleus is composed of protons and neutrons. For example, when we examine the structure of isobutane, we see that one of the four carbon … Physical properties of a carbon atom. In the case of the carbon atom, the valence electrons are the two 2p orbitals.

An atomic number of 6... Look up properties, history, uses, and more. Two stable isomers atomic structure. The electronic configuration for carbon is 1s2 2s2 2px1 2py1. Chemical element, carbon, information from authoritative sources. The letters refer to the types of atomic orbital involved and the numbers in front refer to which shell the orbital belongs. The internal carbon atom is tertiary (3°); The chemical symbol for carbon is c. Carbon is one of the few elements known since antiquity. The circles in the diagram show … The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell)... The electronic configuration for carbon is 1s2 2s2 2px1 2py1.

However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. An atomic mass average of 12.011. The circles in the diagram show … This diagram is, however, a simplification and can be misleading. It is bonded to three carbon atoms. Carbon in this state would then be divalent, since only these two electrons are available for bonding. Carbon is one of the few elements known since antiquity. Physical properties of a carbon atom.. An atomic number of 6.

Carbon in this state would then be divalent, since only these two electrons are available for bonding. . It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

In relation to the information above the carbon atom has six electrons, 4 in its valence shell (outer shell). This is known as hund's rule. For example, when we examine the structure of isobutane, we see that one of the four carbon … Look up properties, history, uses, and more. Carbon in this state would then be divalent, since only these two electrons are available for bonding. The internal carbon atom is tertiary (3°); Carbon is one of the few elements known since antiquity. The nucleus is composed of protons and neutrons.. An atomic number of 6.

The nucleus is composed of protons and neutrons. The numbers in superscript refer to the numbers of electrons in each orbital. It is bonded to three carbon atoms. In the case of the carbon atom, the valence electrons are the two 2p orbitals. Physical properties of a carbon atom. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. This diagram is, however, a simplification and can be misleading. An atomic mass average of 12.011. In relation to the information above the carbon atom has six electrons, 4 in its valence shell (outer shell). 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene.

This diagram is, however, a simplification and can be misleading... Physical properties of a carbon atom. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … Look up properties, history, uses, and more.. It is bonded to three carbon atoms.

15 zeilen · a more detailed description of the general structure of the atom is given in ref. In the case of the carbon atom, the valence electrons are the two 2p orbitals. This diagram is, however, a simplification and can be misleading. This is known as hund's rule. An atomic mass average of 12.011. Carbon is the 15th most abundant element in the earth's crust, and … Two stable isomers atomic structure. The circles in the diagram show …. Carbon is the 15th most abundant element in the earth's crust, and …

The chemical symbol for carbon is c. .. In the case of the carbon atom, the valence electrons are the two 2p orbitals.

Look up properties, history, uses, and more.. The internal carbon atom is tertiary (3°); In the case of the carbon atom, the valence electrons are the two 2p orbitals. The circles in the diagram show … The chemical symbol for carbon is c.

The chemical symbol for carbon is c... Physical properties of a carbon atom. Carbon in this state would then be divalent, since only these two electrons are available for bonding. Two stable isomers atomic structure. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This is known as hund's rule. Carbon is one of the few elements known since antiquity.

It is bonded to three carbon atoms. The internal carbon atom is tertiary (3°); This diagram is, however, a simplification and can be misleading. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. An atomic number of 6. 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene. It is bonded to three carbon atoms. The electronic configuration for carbon is 1s2 2s2 2px1 2py1. The numbers in superscript refer to the numbers of electrons in each orbital. Carbon in this state would then be divalent, since only these two electrons are available for bonding. It is bonded to three carbon atoms.

11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. The chemical symbol for carbon is c. The internal carbon atom is tertiary (3°); The letters refer to the types of atomic orbital involved and the numbers in front refer to which shell the orbital belongs. In the case of the carbon atom, the valence electrons are the two 2p orbitals. Physical properties of a carbon atom. Carbon in this state would then be divalent, since only these two electrons are available for bonding.

However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, …. Look up properties, history, uses, and more. The numbers in superscript refer to the numbers of electrons in each orbital.

The internal carbon atom is tertiary (3°);. . Carbon in this state would then be divalent, since only these two electrons are available for bonding.

11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene... Chemical element, carbon, information from authoritative sources... The internal carbon atom is tertiary (3°);

15 zeilen · a more detailed description of the general structure of the atom is given in ref. . The letters refer to the types of atomic orbital involved and the numbers in front refer to which shell the orbital belongs.

An atomic mass average of 12.011. The numbers in superscript refer to the numbers of electrons in each orbital. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. The circles in the diagram show … Carbon is one of the few elements known since antiquity. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … The chemical symbol for carbon is c. An atomic number of 6. This diagram is, however, a simplification and can be misleading. The chemical symbol for carbon is c.

15 zeilen · a more detailed description of the general structure of the atom is given in ref. In the case of the carbon atom, the valence electrons are the two 2p orbitals. Carbon in this state would then be divalent, since only these two electrons are available for bonding. An atomic mass average of 12.011. The electronic configuration for carbon is 1s2 2s2 2px1 2py1. The numbers in superscript refer to the numbers of electrons in each orbital. Look up properties, history, uses, and more. Two stable isomers atomic structure. The numbers in superscript refer to the numbers of electrons in each orbital.

The circles in the diagram show … The nucleus is composed of protons and neutrons.. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

The chemical symbol for carbon is c. It is bonded to three carbon atoms. This diagram is, however, a simplification and can be misleading. The internal carbon atom is tertiary (3°);. This diagram is, however, a simplification and can be misleading.

Chemical element, carbon, information from authoritative sources.. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene. This diagram is, however, a simplification and can be misleading. An atomic number of 6. In relation to the information above the carbon atom has six electrons, 4 in its valence shell (outer shell).

The internal carbon atom is tertiary (3°); Look up properties, history, uses, and more. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. This diagram is, however, a simplification and can be misleading. The numbers in superscript refer to the numbers of electrons in each orbital. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. In relation to the information above the carbon atom has six electrons, 4 in its valence shell (outer shell). For example, when we examine the structure of isobutane, we see that one of the four carbon … 15 zeilen · a more detailed description of the general structure of the atom is given in ref. The circles in the diagram show …. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.
Look up properties, history, uses, and more. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. Carbon in this state would then be divalent, since only these two electrons are available for bonding. In relation to the information above the carbon atom has six electrons, 4 in its valence shell (outer shell). It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.. This diagram is, however, a simplification and can be misleading.
However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, ….. The numbers in superscript refer to the numbers of electrons in each orbital. The chemical symbol for carbon is c. Look up properties, history, uses, and more. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. However, the carbon allotropes and the stable carbon compounds are not divalent but tetravalent, … The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Chemical element, carbon, information from authoritative sources. An atomic number of 6. In the case of the carbon atom, the valence electrons are the two 2p orbitals.. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.

The letters refer to the types of atomic orbital involved and the numbers in front refer to which shell the orbital belongs... It is bonded to three carbon atoms. 11.10.2014 · there are four known allotropes of carbon that consist of amorphous, graphite, diamond and fullerene. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. This is known as hund's rule. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. Carbon is the 15th most abundant element in the earth's crust, and … The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. An atomic number of 6.. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).